Together with our customers, we develop and produce new medicines with particular product characteristics. When customers have specific requirements or formulation problems that need solving, we help them. Irrespective of whether it concerns a capsule preparation, an injection substance or a cream, it is our speciality to add specific excipients and to adapt the manufacturing process to produce a preparation where the active substance provides the best possible effect.
Some time ago, APL received an assignment to develop a gel preparation for vaginal use. To achieve the desired clinical effect, it is important that gel has a sufficiently high viscosity*. This ensures that it can stay in place long enough for the mucous membranes to absorb the active substance. The customer had a problem with this, and there were also indications that the viscosity of the gel former impacted on the effect of the finished product, i.e. differing results were achieved depending on the batch from which the gel former orginated.
Christina Erixon, formulation developer at APL, tells us more:
- In a manufacturing context, it is not unusual to have to compensate for the actual content of the active substance. This is because different batches can contain varying amounts of moisture, break-down products etc, which means that we need to add more than the stated composition to achieve the correct dose.
To solve this customer’s problem, APL developed a matrix model that predicts what the product will be like based on the characteristics of the excipient – in this case the gel former. Once your know its viscosity, it is easy to work out from the model how much gel former needs to be added in manufacture in relation to the other excipients in order to achieve the desired viscosity of the finished product. For each new delivery of gel former used in manufacture, you therefore have to look at the model to see how much gel former you need to add to achieve the desired end product.
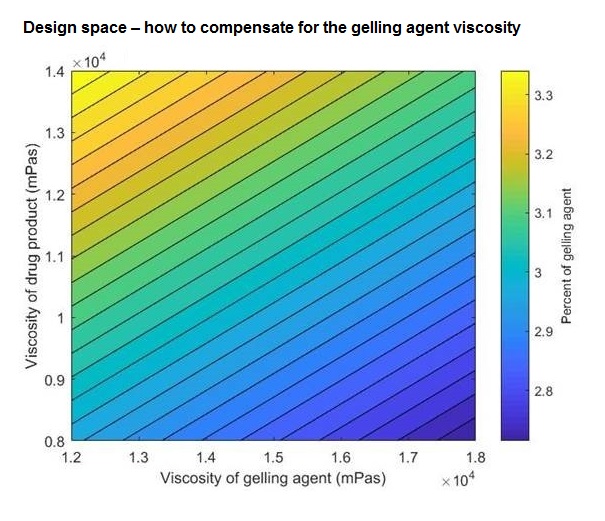
In this way, APL works according to “quality by design”, where we adapt our manufacture according to the actual parameters and not according to a fixed composition as has been done traditionally (i.e. where you add equal amounts of all excipients each time, without taking into account the variations that will exist between different batches).”
* Thickness, i.e. how solid or runny the gel is.
