- Home
- In English
- About APL
About APL
We are one of Europe’s largest manufacturers of extemporaneous medicines and a leading contract manufacturer within Life Science
Our operation
Our task is to develop and manufacture individually adapted medicines for patients with special needs, such as children who need specific strengths or flavours, and persons suffering from various over-sensitivities. The operation is carried out in dialogue and close collaboration with specialists, county councils and public agencies, to ensure that patients get access to the correct treatment.
APL also carries out development and contract manufacture of medicines on behalf of biotechnology and pharmaceutical companies. We also have resources to develop, manufacture and analyse products such as specialist medicines and other niche products.

Our history
The Swedish pharmacy sector was nationalised in the early 1970s, and Apoteksbolaget AB was formed. This company included Apotek Produktion & Laboratorium AB (APL), which was tasked to provide prescribers and therefore patients with extemporaneous medicines, or specially manufactured preparations for individually adapted treatment. In conjunction with the deregulation of the pharmacy market in 2009, it was no longer permissible for pharmacies to produce medicines simultaneously, and therefore APL was disconnected from Apoteket AB in June 2010.

Vision, mission and goals

Our vision
We make a difference for patients with special needs
Our mission
We develop and manufacture extemporaneous and orphan drugs as part of a social mission and on behalf of other pharmaceutical companies, to create the greatest possible patient benefit.

Our goals
- We aim to be a sustainable and efficient supplier
- We aim to be competitive and profitable supplier
- We aim to be an attractive employer
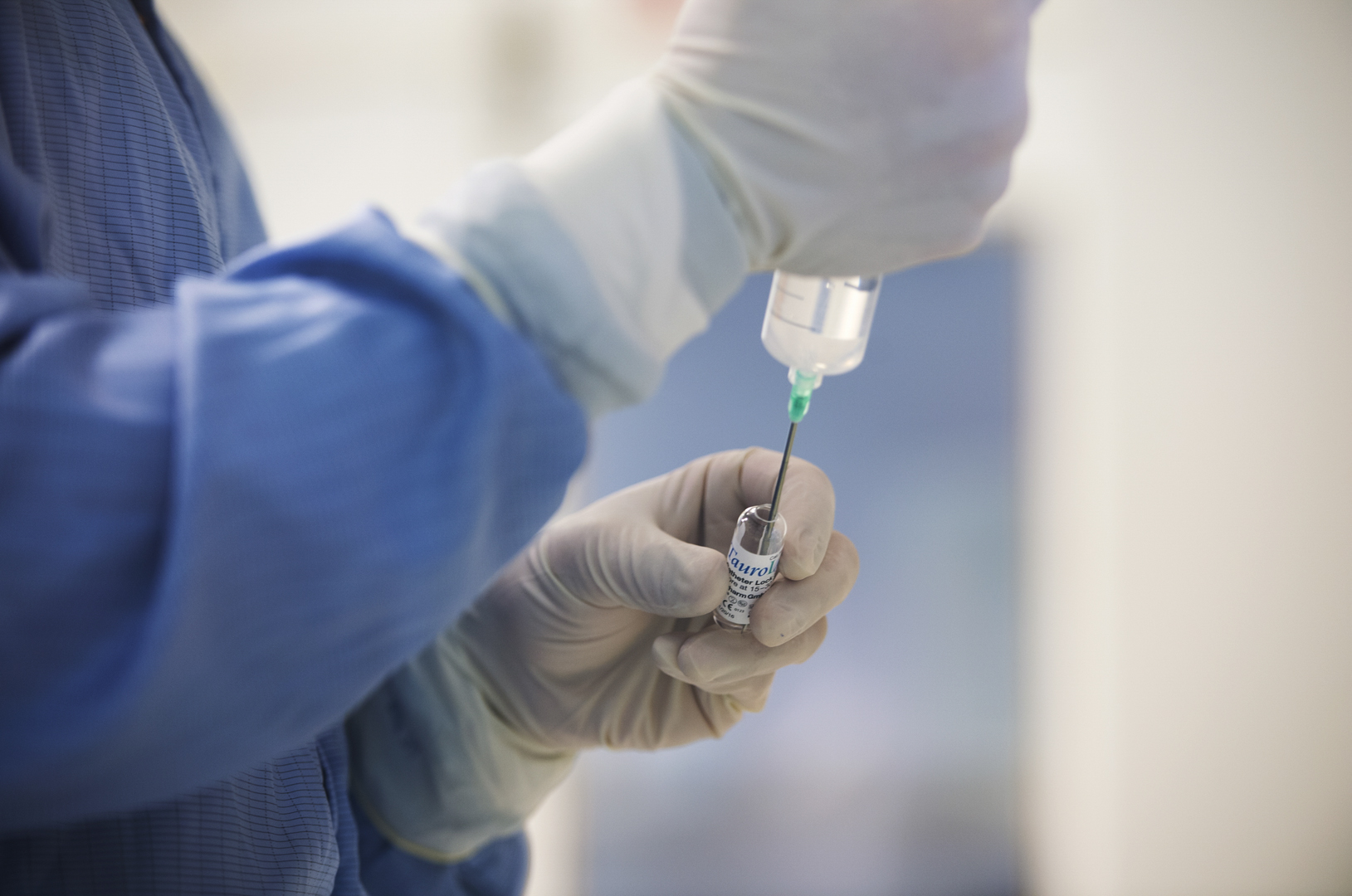
When a life is at stake, every minute counts
View an intensive and carefully controlled process in sterile manufacturing at APL.

Sustainable development
We constantly analyse our activities, products and services in order to identify improvement measures within quality, environment and in the social sphere.

Work for us
We need more skilled, solution-oriented and responsible empolyees and we can offer opportunities for development in an exciting industry.
APL in figures
780 mkr
Net turnover
2 000
Extemporaneous medicines
484
Number of employees


